Physics 9702 Doubts | Help Page 187
Question 912: [Oscillations
> Simple harmonic motion]
A student sets out to investigate
the oscillation of a mass suspended from the free end of a spring, as
illustrated in Fig.1.
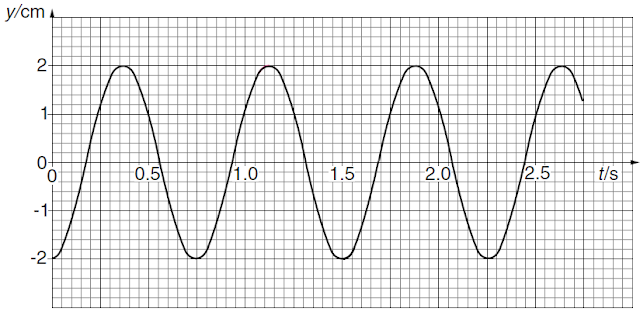
The mass is pulled downwards and
then released. The variation with time t of the displacement y of the mass is
shown in Fig.2.
(a) Use information from Fig.2
(i) to explain why the graph
suggests that the oscillations are undamped,
(ii) to calculate angular frequency
of the oscillations,
(iii) to determine maximum speed of
the oscillating mass.
(b)
(i) Determine resonant frequency f0
of the mass-spring system.
(ii) The student finds that if short
impulsive forces of frequency ½ f0 are impressed on the mass-spring
system, a large amplitude of oscillation is obtained. Explain this observation.
Reference: Past Exam Paper – November 2002 Paper 4 Q3
Solution 912:
(a)
(i) It has a constant amplitude.
(ii)
Period T = 0.75s
Angular frequency ω = 2π / T
= 8.4 rad s–1
(iii)
EITHER use of gradient OR v = ωy0
{y0 is the amplitude}
Maximum speed v = {8.4 × 0.02 =} 0.168 m s–1
(b)
(i) Resonant frequency f0
{= 1/T = 1/0.75} = 1.3Hz
(ii) At ½ f0, a ‘pulse’
is provided to the mass on alternate/some oscillations. So, the ‘pulses’ build up
the amplitude.
Question 913: [Current
of Electricity]
(a) Distinguish between potential difference (p.d.) and electromotive
force (e.m.f.) in terms of energy transformations.
(b) Two cells A and B are connected in series with a resistor R of
resistance 5.5 Ω, as shown in Fig.1.
Cell A has e.m.f. 4.4 V and internal
resistance 2.3 Ω. Cell B has e.m.f. 2.1 V and internal resistance 1.8 Ω.
(i) State Kirchhoff’s second law.
(ii) Calculate current in the
circuit.
(iii) On Fig.1, draw an arrow to
show the direction of the current in the circuit. Label this arrow I.
(iv) Calculate
1. p.d. across resistor R,
2. terminal p.d. across cell A,
3. terminal p.d. across cell B.
Reference: Past Exam Paper – November 2011 Paper 23 Q4
Solution 913:
(a)
Potential difference (p.d.) is the
energy transformed from electrical to other forms, per unit charge.
Electromotive force (e.m.f.) is the energy
transformed from other forms to electrical per unit charge.
(b)
(i) Kirchhoff’s second law states
that the sum of e.m.f.s (in a closed circuit) is equal to the sum of potential
differences.
(ii)
4.4 – 2.1 = I × (1.8 + 5.5 + 2.3)
Current I = 0.24 A
(iii) The arrow (labelled) I should
be shown anticlockwise.
(iv)
1. V = I × R = 0.24 × 5.5 = 1.3(2) V
2. VA = 4.4 – (I × 2.3) =
3.8(5) V
{Voltage loss in cell A = I
× 2.3}
3.
EITHER VB = 2.1 + (I ×
1.8) OR VB
= 3.8 – 1.3
{Here, we need to add (I ×
1.8) to the e.m.f. of cell B because the current is flowing towards the
positive terminal of cell B.}
VB = 2.5(3) V
Question 914: [Dynamics
> Moments]
Which pair of forces acts as a
couple on the circular object?
Reference: Past Exam Paper – November 2008 Paper 1 Q14
Solution 914:
Answer: A.
A couple consists of 2 forces, equal
in magnitude. But acting in opposite directions, so that they produce a
resultant turning effect. Thus, these 2 forces produces a resultant moment,
while the resultant force is zero.
Choice B: both forces are in the same
direction + their magnitudes are not equal
Choice C: different magnitudes
Choice D: directions are not
opposite to each other
Question 915: [Kinetic
theory of Gases]
(a)
(i) State the basic assumption of
the kinetic theory of gases that leads to the conclusion that potential energy
between the atoms of an ideal gas is zero.
(ii) State what is meant by the internal
energy of a substance.
(iii) Explain why an increase in
internal energy of an ideal gas is directly related to a rise in temperature of
the gas.
(b) A fixed mass of an ideal gas undergoes a cycle PQRP of changes as
shown in Fig.1.
(i) State the change in internal
energy of the gas during one complete cycle PQRP.
(ii) Calculate work done on the gas
during the change from P to Q.
(iii) Some energy changes during the
cycle PQRP are shown in Fig.2.
Complete Fig.2 to show all of the
energy changes.
Reference: Past Exam Paper – November 2010 Paper 41 & 42 Q2
Solution 915:
A fixed mass of an ideal gas undergoes a cycle PQRP of changes as shown in Fig. 2.1.






For 42/O/N/10 Q.2(b)(iii) How to get the values of the energy changes in the table? Which method should I use?
ReplyDeleteThe method has now been included
DeletePlease consider answering ALL of the following questions before October:
ReplyDelete4/O/N/02 Q.5(b),Q.6(c)(i)
6/O/N/02 Q.11(a)(b)
6/O/N/03 Q.9
04/M/J/04 Q.4(c),Q.8(a),(b)(i),(ii)1.
06/M/J/04 Q.9(b)(iii),Q.11(b)
06/O/N/04 Q.3(b)(i)1.,Q.4(a),Q.6(b),Q.8(a)(i)
04/M/J/05 Q.7(a)
06/O/N/05 Q.8(b),Q.10(a)
04/M/J/06 Q.6(a),(c),Q.7(b)
06/M/J/06 Q.14(b)
04/O/N/06 Q.3(c)
06/O/N/06 Q.3(b)
05/M/J/07 Q.2(d)
04/O/N/07 Q.7(b)(i),(c),Q.10(c)
04/M/J/08 Q.3(c)(ii),Q.5(b),Q.9(b),Q.11(a)(ii)
04/O/N/08 Q.7(c)
04/M/J/09 Q.10(c)(ii),Q.11(b)(iii)
41/O/N/09 Q.6(a),(b)(i),Q.10
42/O/N/09 Q.5(a),Q.7(b)(ii)
41/M/J/10 Q.6(a),Q.7(a)
42/M/J/10 Q.6(a)(ii)
51/M/J/10 Q.2(d)
42/O/N/10 Q.3(c)
For 04/M/J/04 Q.4(c), see solution 916 at
Deletehttp://physics-ref.blogspot.com/2015/08/physics-9702-doubts-help-page-188.html
2004 O/N paper01 Q15
ReplyDeletethe kinetic energy of a particle is increased by a factor of 4,
by what factor does the speed increase?
See solution 67 at
Deletehttp://physics-ref.blogspot.com/2014/11/physics-9702-doubts-help-page-12.html
Hi,
ReplyDeleteWhy is the change in internal energy during one complete cycle in pqrp is zero?
From the first law of thermodynamics, the internal energy depends on the state of the system, that is, its temperature, pressure and volume.
ReplyDeletein a complete cycle, the final state of the system (P, V and T) and the same as its initial state. So, there is no change.
change in internal energy = 0
Can anyone tell me how is the change in internal energy of the gas during one complete cycle PQRP is zero?
ReplyDeleteyou need to provide the year and paper
DeleteExplain what is meant by internal resistance of a cell.
ReplyDelete(b) A cell has an emf of 1.52V and an internal resistance of 0.450 ohms. Fig. 4.1 shows the cell connected to a variable resistor R.
ReplyDeleteI cant find this qs anywhere and i need the answers to this qs. Pls help
When calculating work done from R to P why does W=pV not provide the correct answer?
ReplyDeletesome details have been added.
Deletehope it helps